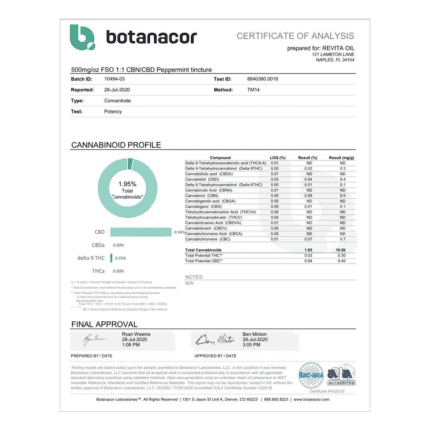
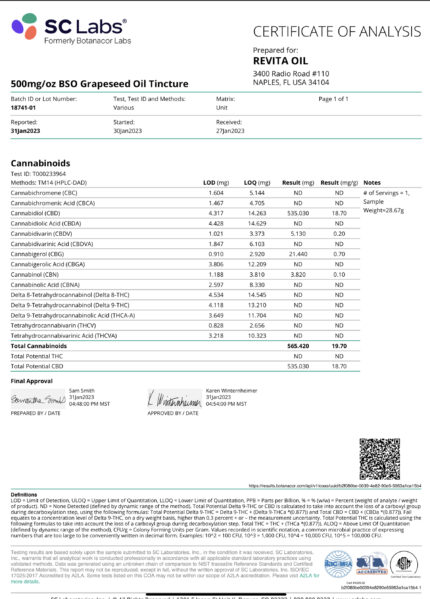
CBD Products By Revita Oil
CBD Oil
Organic • Full Spectrum
Cannabidiol (CBD) is one of the most medicinal compounds in the cannabis plant and it’s been utilized as medicine for thousands of years

We are a team of passionate natural health advocates and are dedicated to providing the absolute best CBD to our clients
Featured CBD Products


NEW! Delta 8
Say HELLO! To Our Custom Formulated Delta 8 CBD
Delta-8 tetrahydrocannabinol is a cannabinoid found in the Hemp plant.